Oxysterols are oxidized derivatives of cholesterol that are present at low levels yet have broad biological significance. Once regarded mainly as intermediates of sterol metabolism, they are now understood as signaling molecules involved in transcriptional control, immune-cell positioning, inflammatory regulation, and disease development. This shift in understanding has brought oxysterols to the forefront of research in cardiovascular disease, cancer, neuroscience, and biomarker discovery. Their analysis, however, remains technically demanding because many oxysterols are chemically labile and structurally similar. This article outlines how oxysterols are formed, how they function, why they matter in disease, and how they can be measured reliably.
1. What Are Oxysterols? Definition, Formation, and Major Types
Oxysterols are oxygenated derivatives of cholesterol that retain the sterol backbone while acquiring one or more additional oxygen-containing groups, including hydroxyl, ketone, epoxide, or carboxyl moieties. Oxidation can occur on the steroid ring or the aliphatic side chain, and the site of modification strongly influences receptor binding, metabolic fate, and biological activity (Kovač et al., 2019; Griffiths & Wang, 2020).
Oxysterols arise through two main pathways: regulated enzymatic biosynthesis and non-enzymatic autoxidation:
- Enzymatic formation is highly regulated and structurally specific, and is mediated primarily by cytochrome P450 (CYP) enzymes and related oxidases. These pathways generate defined oxysterol species that serve as endogenous ligands for nuclear receptors and as regulators of cholesterol metabolism, immune signaling, and cellular homeostasis.
- Non-enzymatic formation, by contrast, results from the reaction of cholesterol with reactive oxygen species (ROS) and other oxidizing conditions. This process is typically associated with oxidative stress, inflammation, aging, and disease-related tissue damage, and often gives rise to oxysterols with cytotoxic or pro-inflammatory properties.
Several oxysterol species are especially important because of their well-defined metabolic origins and disease relevance:
- 24S-hydroxycholesterol (24S-HC): Predominantly synthesized in the brain by CYP46A1, 24S-HC serves as a major route for the elimination of excess cholesterol from neurons.
- 25-hydroxycholesterol (25-HC): Produced by cholesterol 25-hydroxylase (CH25H), especially in macrophages and other immune cells, 25-HC functions as an important antiviral effector and immune modulator.
- 27-hydroxycholesterol (27-HC): Generated by CYP27A1 in the liver and peripheral tissues, 27-HC is one of the major circulating oxysterols and has also been described as a selective estrogen receptor modulator (SERM).
- 7alpha-hydroxycholesterol (7alpha-HC): Formed by CYP7A1 in the liver, 7alpha-HC is a key intermediate in the classical pathway of bile acid synthesis.
- 7-ketocholesterol (7-KC): Produced mainly through non-enzymatic oxidation, 7-KC is a major cytotoxic component of oxidized low-density lipoprotein (oxLDL) and is widely associated with oxidative injury and vascular pathology.
- 7alpha,25-dihydroxycholesterol (7alpha,25-diHC): This dihydroxylated oxysterol acts as a highly specific chemotactic ligand for EBI2/GPR183 and plays an essential role in guiding immune-cell positioning within lymphoid tissues.
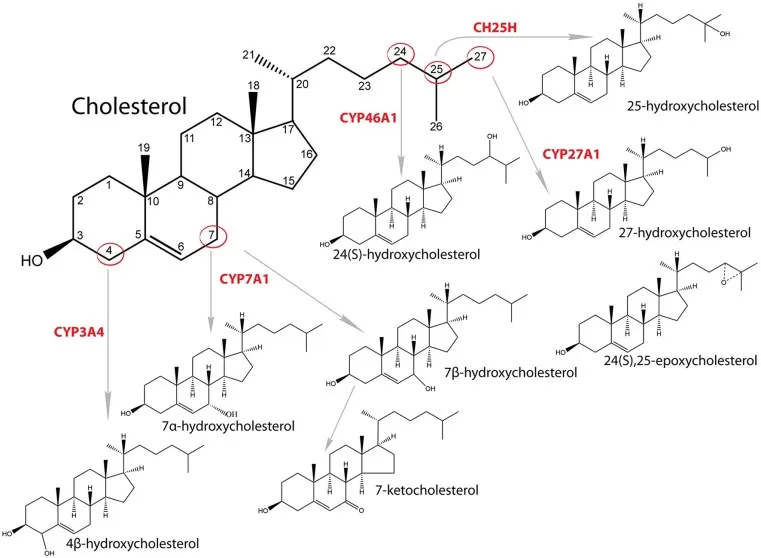
Figure 1. Synthesis and structure of major oxysterols. Image adapted from Kovač et al. (2019), Frontiers in endocrinology.
2. Oxysterol Biosynthesis, Metabolism, and Clearance
Oxysterol abundance in tissues and biofluids reflects a dynamic balance among biosynthesis, downstream metabolism, transport, and clearance. That balance is highly tissue dependent, which is why the same oxysterol can carry different biological meaning depending on the sample type and disease context.
2.1 Tissue-Specific Biosynthesis
The liver is a major site of oxysterol formation because it houses key enzymes involved in cholesterol catabolism and bile acid synthesis. CYP7A1 initiates the classic bile acid pathway through 7alpha-hydroxylation, while CYP27A1 contributes to side-chain oxidation in both hepatic and extrahepatic tissues. Outside the liver, oxysterol production becomes more functionally specialized. In the central nervous system, CYP46A1 converts cholesterol to 24S-HC, enabling excess brain cholesterol to cross the blood-brain barrier in an oxidized form. In macrophages and other immune cells, CH25H expression can be induced during inflammatory and interferon-driven responses, leading to increased 25-HC production. These examples illustrate that oxysterol synthesis is not a generic byproduct of cholesterol metabolism, but a regulated process embedded in tissue physiology (Griffiths & Wang, 2020; Alavi et al., 2023; Zhang et al., 2023).
2.2 Downstream Metabolism and Turnover
After formation, many oxysterols undergo additional enzymatic conversion into bile acids, sulfated sterols, glucuronidated metabolites, or other downstream products (Yamauchi & Rogers, 2018). This rapid turnover is biologically important because it prevents prolonged accumulation of potent signaling lipids while allowing transient regulatory pulses. It also complicates data interpretation: circulating concentrations may not directly represent local tissue production, and a measured oxysterol can reflect both upstream synthesis and downstream clearance efficiency. In practice, oxysterol datasets are most informative when interpreted alongside sample type, disease context, and related lipid pathways rather than as isolated concentrations (Griffiths & Wang, 2020).
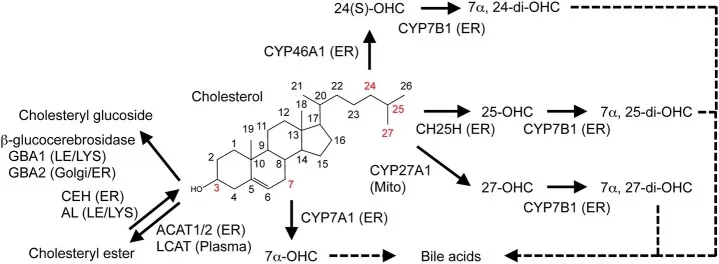
Figure 2. Metabolic conversion of cholesterol. Enzymatic conversion of cholesterol into oxysterols, cholesteryl ester, and cholesteryl glucoside is shown. Image adapted from Yamauchi & Rogers (2018), Frontiers in endocrinology.
3. How Oxysterols Signal: Key Molecular Mechanisms
The biological significance of oxysterols lies in their ability to act as signaling molecules rather than passive oxidation products. Their effects span transcriptional regulation, intracellular sterol sensing, chemotactic guidance, and context-dependent modulation of developmental and hormone-related pathways.
3.1 LXR-Mediated Transcriptional Regulation
Several oxysterols function as endogenous ligands of liver X receptors (LXRs), nuclear receptors that integrate sterol status with transcriptional programs controlling cholesterol efflux, lipid homeostasis, and inflammatory responses. Oxysterol-activated LXR signaling can induce transporters such as ABCA1 and ABCG1, thereby promoting cholesterol export, while also influencing fatty acid metabolism and immune-cell function. Through this mechanism, oxysterols help convert changes in cholesterol burden into coordinated gene-expression responses (Griffiths & Wang, 2020).
3.2 EBI2/GPR183 and Immune-Cell Positioning
A distinct signaling axis is centered on 7alpha,25-diHC, the best-characterized endogenous ligand for EBI2, also known as GPR183. This receptor controls the positioning of B cells, dendritic cells, and other immune populations within lymphoid tissues by sensing local oxysterol gradients. The identification of 7alpha,25-diHC as a potent EBI2 agonist established oxysterols as genuine immune-guidance molecules rather than merely metabolic intermediates. This pathway remains one of the clearest examples of oxysterol signaling with direct spatial consequences for adaptive immunity (Hannedouche et al., 2011).
3.3 SREBP-SCAP-INSIG Sterol Sensing
Oxysterols also participate in post-translational sterol feedback through the SREBP-SCAP-INSIG system. In this context, specific oxysterols help restrain sterol regulatory element-binding protein activation when cholesterol availability is sufficient, thereby reducing endogenous cholesterol biosynthesis. This feedback loop operates more rapidly than classical transcriptional adaptation and is central to intracellular sterol homeostasis (Griffiths & Wang, 2020).
3.4 Other Oxysterol Signaling Pathways
Beyond LXR, EBI2, and SREBP-associated sensing, certain oxysterols influence Smoothened-dependent Hedgehog signaling and estrogen receptor-related biology in a context-dependent manner. These interactions are especially relevant in development, cancer, and tissue remodeling, where oxysterol composition can shape signaling output without acting as a universal on-or-off switch. The same oxysterol may therefore exert different effects depending on receptor repertoire, metabolic background, and cell type (Griffiths & Wang, 2020; Vini et al., 2022).
4. Oxysterols in Disease Mechanisms and Translational Research
Because oxysterols connect cholesterol metabolism with signaling, they are increasingly studied as mechanistic mediators and candidate biomarkers across multiple disease areas. Their roles are not uniform across tissues, and the same molecule may be protective in one setting and pathogenic in another, which makes context a critical part of interpretation.
4.1 Cardiovascular Disease and Atherosclerosis
In cardiovascular research, oxysterols are strongly linked to oxidized lipoproteins, foam-cell biology, endothelial dysfunction, and plaque progression. Among them, 7-KC is particularly important because it is formed during cholesterol autoxidation, accumulates in pathological settings, and has been associated with oxidative stress, cytotoxicity, inflammatory signaling, and lysosomal dysfunction. These features make 7-KC a useful example of how non-enzymatic oxysterols can contribute to vascular pathology rather than simply marking it (Gajendran et al., 2025).
4.2 Cancer Biology and the Tumor Microenvironment
Cancer biology has highlighted the importance of 27-HC, especially in hormone-responsive settings. 27-HC can modulate estrogen receptor signaling and has been implicated in breast cancer biology, while circulating 27-HC has also been evaluated epidemiologically in relation to breast cancer risk. These findings have strengthened interest in the cholesterol-oxysterol axis as a bridge between systemic lipid metabolism and tumor behavior. More broadly, tumor-associated oxysterols may help shape immune-cell recruitment and the immunoregulatory tone of the tumor microenvironment (Vini et al., 2022; DeRouen et al., 2023).
4.3 Neurological Disease and Brain Cholesterol Turnover
The brain has a relatively autonomous cholesterol economy, making oxysterols especially informative in neurological disease. CYP46A1-mediated formation of 24S-HC is a major route for cholesterol elimination from the central nervous system, and altered 24S-HC dynamics have therefore been investigated in neurodegenerative disorders and demyelinating conditions. Although interpretation depends on disease stage, sample type, and neuronal loss, 24S-HC remains one of the most biologically grounded oxysterol markers of brain cholesterol turnover (Alavi et al., 2023; Tripodi et al., 2024).
4.4 Infection, Inflammation, and Immune Regulation
Oxysterol signaling is also highly relevant to host defense. 25-HC is induced in interferon-responsive settings and can restrict viral infection through mechanisms linked to membrane composition, cholesterol accessibility, and broader regulation of antiviral signaling. At the same time, oxysterol gradients, particularly those involving 7alpha,25-diHC, support immune-cell localization during adaptive responses. Together, these functions place oxysterols at the intersection of innate defense and immune organization (Hannedouche et al., 2011; Zhang et al., 2023).
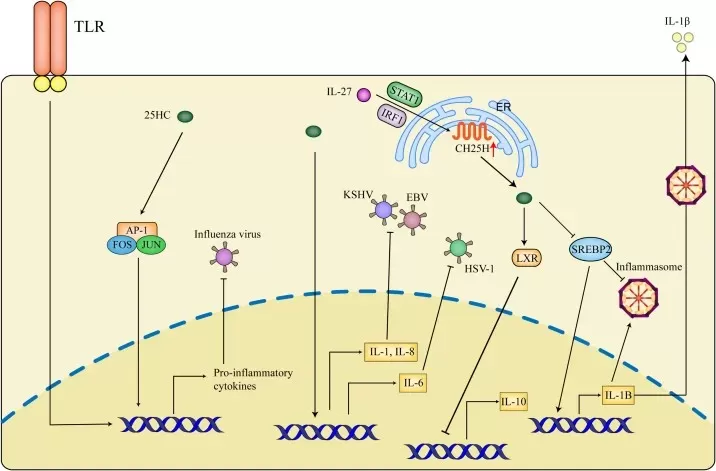
Figure 3. Inflammatory effects of 25-HC in antiviral activities. Image adapted from Zhang et al. (2023), Frontiers in immunology.
5. Analytical Challenges and Strategies for Oxysterol Profiling
Biological relevance alone does not make oxysterols easy to study. In practice, oxysterol measurement is among the more demanding tasks in lipid analysis because low abundance, structural similarity, and oxidative artifact formation all interfere with reliable quantification.
5.1 Why Oxysterol Analysis Is Technically Challenging
Many oxysterols are present only at trace to nanomolar concentrations, yet they coexist with overwhelming amounts of cholesterol and other lipids. In addition, numerous analytes differ only by the position or stereochemistry of a single oxygen-containing group. This combination of low abundance and high isomeric complexity means that generic lipid workflows often lack the selectivity required for confident oxysterol assignment (Kømurcu et al., 2024; Roumain & Muccioli, 2025).
5.2 Pre-Analytical Control and Sample Handling
Accurate oxysterol analysis depends heavily on sample collection, storage, extraction, and handling conditions. During these stages, cholesterol is susceptible to ex vivo oxidation, particularly when samples are exposed to air, light, inappropriate temperatures, or prolonged processing times. This can generate artifactual oxysterols that distort the endogenous profile and reduce confidence in downstream biological interpretation. For this reason, oxysterol workflows typically require rapid freezing, tightly controlled handling procedures, avoidance of repeated freeze-thaw cycles, and the use of antioxidants where needed. Strong pre-analytical control helps preserve sample integrity and supports accurate, reproducible quantification of oxysterols (Griffiths & Wang, 2020; Roumain & Muccioli, 2025).
5.3 LC-MS/MS for Reliable Oxysterol Profiling
Liquid chromatography-tandem mass spectrometry (LC-MS/MS) has become the preferred platform for oxysterol profiling because it combines sensitivity, molecular specificity, and compatibility with targeted quantification. Reliable workflows depend on more than the mass spectrometer alone. Chromatographic separation is essential for resolving isomeric species, sample cleanup is needed to reduce interference from abundant cholesterol, and derivatization may be used for selected compounds to improve ionization efficiency and sensitivity. Stable-isotope internal standards are also central to robust quantification because they help correct for extraction losses, matrix effects, and analytical variability. Recent method-development studies have shown that carefully optimized LC-MS/MS workflows can support broad, quantitative profiling of oxysterols across diverse biosamples (Kømurcu et al., 2024; Roumain & Muccioli, 2025).
5.4 Targeted vs Untargeted Strategies for Oxysterol Studies
Untargeted lipidomics remains valuable for exploratory studies, especially when oxysterol biology is being considered within a broader lipidome context. However, oxysterols are often underrepresented in untargeted datasets because they ionize less efficiently than many abundant lipid classes and may fall below confident detection thresholds. When oxysterols are a primary biological focus, targeted assays are generally more informative because they offer higher sensitivity, better isomer discrimination, and stronger quantitative performance. The best platform choice therefore depends on study intent: untargeted workflows for discovery-oriented breadth, and targeted LC-MS/MS for mechanistic, translational, or biomarker-driven questions (Kømurcu et al., 2024; Roumain & Muccioli, 2025).
MetwareBio: Your Trusted Partner for Oxysterol Analysis
Oxysterols are important regulators of cholesterol metabolism, immune signaling, and disease biology. Accurate and reliable quantification of these molecules can support mechanistic studies and biomarker discovery in inflammation, cancer, cardiovascular disease, and neurobiology. For researchers who need targeted oxysterol analysis, MetwareBio provides a comprehensive solution. Our LC-MS/MS based Steroid Hormones Targeted Metabolomics service can quantify key oxysterols such as 24-hydroxycholesterol, 25-hydroxycholesterol, 7-ketocholesterol, 7alpha,25-dihydroxycholesterol, and 7alpha,27-dihydroxycholesterol, in addition to cholesterol and metabolites across major steroid hormone classes.
The core strengths of this targeted service include:
- Absolute quantification supported by 40 external standards and 23 internal standards
- High sensitivity based on the AB QTRAP 6500+ LC-MS/MS platform, with ng/mL-level detection capability
- Broad coverage spanning core steroid hormones together with oxysterols and related sterol metabolites
If you are interested in oxysterol analysis, please do not hesitate to contact us for further discussion of study design, sample preparation, panel coverage, or other project-specific questions.
Contact UsRead More: Related Guides for Oxysterol and Cholesterol Research
Want to explore the broader metabolic context behind oxysterols? These articles cover cholesterol biology, upstream biosynthetic pathways, and the disease-specific roles of key oxysterol species.
Cholesterol in Health and Disease: Functions, Metabolism, Homeostasis, and Omics Research
Start with a broader view of cholesterol biology, including its metabolism, homeostatic regulation, and relevance to disease and omics research.
The Mevalonate Pathway: Central Hub of Cholesterol Metabolism, Isoprenoid Biosynthesis, and Disease Mechanisms
Learn how the upstream mevalonate pathway supports cholesterol synthesis and shapes the metabolic foundation for sterol-derived signaling molecules such as oxysterols.
25-Hydroxycholesterol (25-HC): Roles in Immunity, Inflammation, and Disease
Explore one of the most important immune-related oxysterols and its roles in antiviral defense, inflammatory regulation, and disease mechanisms.
27-Hydroxycholesterol in Cancer: How 27-HC Links Lipid Metabolism and Hormone Signaling
See how 27-HC connects cholesterol metabolism with estrogen receptor signaling and tumor biology, especially in hormone-related cancers.
References
- Griffiths, W. J., & Wang, Y. (2020). Oxysterols as lipid mediators: Their biosynthetic genes, enzymes and metabolites. Prostaglandins & other lipid mediators, 147, 106381. https://doi.org/10.1016/j.prostaglandins.2019.106381
- Kovač, U., Skubic, C., Bohinc, L., Rozman, D., & Režen, T. (2019). Oxysterols and Gastrointestinal Cancers Around the Clock. Frontiers in endocrinology, 10, 483. https://doi.org/10.3389/fendo.2019.00483
- Yamauchi, Y., & Rogers, M. A. (2018). Sterol Metabolism and Transport in Atherosclerosis and Cancer. Frontiers in endocrinology, 9, 509. https://doi.org/10.3389/fendo.2018.00509
- Alavi, M. S., Karimi, G., Ghanimi, H. A., & Roohbakhsh, A. (2023). The potential of CYP46A1 as a novel therapeutic target for neurological disorders: An updated review of mechanisms. European journal of pharmacology, 949, 175726. https://doi.org/10.1016/j.ejphar.2023.175726
- Zhang, J., Zhu, Y., Wang, X., & Wang, J. (2023). 25-hydroxycholesterol: an integrator of antiviral ability and signaling. Frontiers in immunology, 14, 1268104. https://doi.org/10.3389/fimmu.2023.1268104
- Hannedouche, S., Zhang, J., Yi, T., Shen, W., Nguyen, D., Pereira, J. P., ... & Sailer, A. W. (2011). Oxysterols direct immune cell migration via EBI2. Nature, 475(7357), 524–527. https://doi.org/10.1038/nature10226
- Vini, R., Rajavelu, A., & Sreeharshan, S. (2022). 27-Hydroxycholesterol, The Estrogen Receptor Modulator, Alters DNA Methylation in Breast Cancer. Frontiers in endocrinology, 13, 783823. https://doi.org/10.3389/fendo.2022.783823
- Gajendran, T. Y., Ganamurali, N., & Sabarathinam, S. (2025). 7-Ketocholesterol: A pathogenic oxysterol in atherosclerosis and lysosomal storage disorders - Molecular insights and clinical implications. The Journal of steroid biochemistry and molecular biology, 252, 106797. https://doi.org/10.1016/j.jsbmb.2024.106797
- DeRouen, M. C., Yang, J., Li, Y., Franke, A. A., ... & Cheng, I. (2023). Circulating 27-hydroxycholesterol, lipids, and steroid hormones in breast cancer risk: a nested case-control study of the Multiethnic Cohort Study. Breast cancer research : BCR, 25(1), 95. https://doi.org/10.1186/s13058-023-01692-5
- Tripodi, D., Vitarelli, F., Spiti, S., & Leoni, V. (2024). The Diagnostic Use of the Plasma Quantification of 24S-Hydroxycholesterol and Other Oxysterols in Neurodegenerative Disease. Advances in experimental medicine and biology, 1440, 337–351. https://doi.org/10.1007/978-3-031-43883-7_16
- Kømurcu, K. S., Wilson, S. R., & Røberg-Larsen, H. (2024). LC-MS Approaches for Oxysterols in Various Biosamples. Advances in experimental medicine and biology, 1440, 57–71. https://doi.org/10.1007/978-3-031-43883-7_3
- Roumain, M., & Muccioli, G. G. (2025). Development and application of an LC-MS/MS method for the combined quantification of oxysterols and bile acids. Journal of lipid research, 66(1), 100697. https://doi.org/10.1016/j.jlr.2024.100697


