Cholesterol is far more than a structural component of cellular membranes. Its oxidative metabolites—collectively known as oxysterols—are increasingly recognized as potent signaling molecules that govern diverse physiological processes. Once dismissed as mere intermediates of cholesterol catabolism, these compounds are now firmly established as key regulators of cholesterol homeostasis, inflammatory responses, antiviral defense, and even cell fate decisions. Among the myriad oxysterols, 25-Hydroxycholesterol (25-HC) has emerged as a molecule of particular interest due to its unique role at the intersection of metabolism and immunity. Functioning both as a cholesterol "sensor" and a critical effector of interferon signaling, 25-HC exhibits remarkably complex and context-dependent regulatory functions in inflammation, antiviral immunity, tumor immunology, and age-related pathologies. This article aims to provide a comprehensive, evidence-based overview of 25-HC, systematically examining its biosynthetic pathways, physiological functions, and dualistic roles in various disease contexts, thereby offering a valuable resource for researchers in the field.
1. What is 25-Hydroxycholesterol?
25-Hydroxycholesterol, abbreviated as 25-HC and systematically named cholest-5-ene-3β,25-diol, is an oxysterol generated by the hydroxylation of cholesterol and belongs to the oxysterol subclass of cholesterol derivatives.
Structurally, 25-HC is cholesterol modified by the introduction of a hydroxyl group (-OH) at the 25th carbon position of the side chain. It possesses the molecular formula C₂₇H₄₆O₂ and an average molecular weight of 402.65 Da. The introduction of the additional hydroxyl group renders 25-HC significantly more polar than its parent molecule, cholesterol. This enhanced polarity facilitates its transport between intracellular compartments and across membranes, and also allows it to integrate more readily into lipid bilayers, potentially modulating membrane fluidity—a feature crucial for its signal-transducing functions.
Chemically, 25-HC behaves as an extremely weak base based on its predicted pKa, remaining largely neutral under physiological conditions. Its structural architecture comprises several key elements: the canonical cyclopentanoperhydrophenanthrene sterol nucleus, a β-oriented hydroxyl group at C3, a double bond between C5 and C6 (Δ5), and the defining C25 hydroxyl group on the flexible side chain. This unique structure enables 25-HC to retain recognition by cholesterol-handling proteins while simultaneously acquiring the ability to engage oxysterol-specific receptors, thereby executing functions distinct from cholesterol itself.
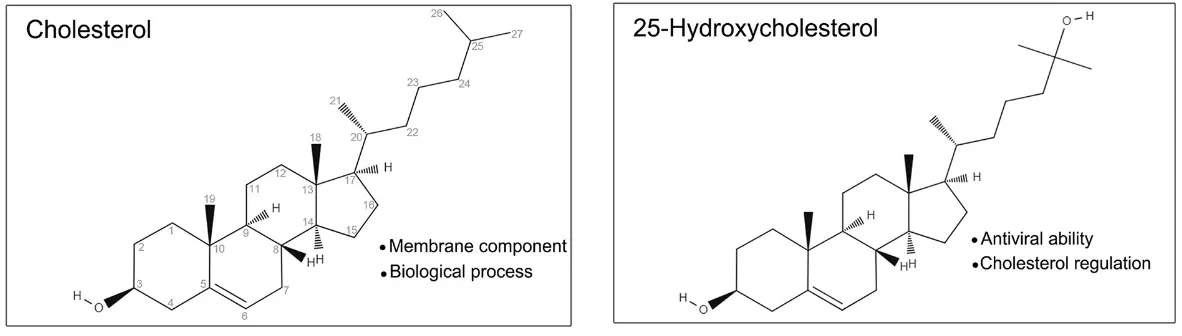
Image reproduced from Zhang et al., 2023, Frontiers in Immunology, licensed under the Creative Commons Attribution 4.0 International License (CC BY 4.0).
2. Biosynthesis and Metabolic Fate of 25-Hydroxycholesterol
Understanding where 25-HC comes from is essential for understanding what it does. Its biosynthesis is not a passive chemical event but a biologically regulated process that changes across tissues, cell types, and disease states. Although non-enzymatic oxidation can generate 25-HC under certain oxidative conditions, the dominant biologically relevant source is enzymatic conversion of cholesterol by cholesterol 25-hydroxylase.
2.1 Enzymatic and Non-Enzymatic Formation of 25-HC
The principal enzymatic source of 25-HC is cholesterol 25-hydroxylase (CH25H), a non-cytochrome P450 enzyme located primarily on the endoplasmic reticulum membrane. CH25H catalyzes side-chain hydroxylation of cholesterol to generate 25-HC. Unlike many sterol hydroxylases, CH25H belongs to a distinct diiron enzyme class, which already signals that its biology is somewhat unusual. Importantly, CH25H is not simply constitutively expressed; it is best understood as an inducible immune-responsive enzyme (Nguyen et al., 2023).
CH25H expression is strongly stimulated by type I interferons, inflammatory cytokines, and pattern-recognition receptor signaling, especially in macrophages, dendritic cells, and other innate immune cells. This is one of the central reasons 25-HC is tightly linked to antiviral immunity and inflammatory regulation. In practical terms, when infection or inflammatory danger signals are sensed, cells can rapidly increase 25-HC production as part of a broader innate defense program (Zhang et al., 2023).
Besides CH25H, minor enzymatic contributions may come from certain cytochrome P450 enzymes such as CYP3A4, although these appear to play a secondary role in most physiological settings. Non-enzymatic formation is also possible through free radical-mediated oxidation of cholesterol, particularly during oxidative stress or food processing, but this route is much more difficult to interpret biologically because it may reflect artifact generation rather than controlled signaling (Nguyen et al., 2023; Romero et al., 2024).
 enzyme-dependent and enzyme-independent._1774834124_WNo_672d350.webp)
Image reproduced from Zhang et al., 2023, Frontiers in Immunology, licensed under the Creative Commons Attribution 4.0 International License (CC BY 4.0).
2.2 Cellular Sources and Downstream Metabolism of 25-HC
Immune cells are among the most important sources of inducible 25-HC, particularly macrophages and dendritic cells, but expression is not restricted to them. Epithelial and endothelial cells can also generate 25-HC under inflammatory conditions, and brain-resident cells such as activated microglia can upregulate CH25H during neuroinflammation. This broader tissue distribution helps explain why 25-HC has been implicated in infection, vascular inflammation, CNS disease, and tumor microenvironment remodeling (Nguyen et al., 2023; Romero et al., 2024).
25-HC is not metabolically static after it is formed. One important downstream conversion is 7α-hydroxylation by CYP7B1, yielding 7α,25-dihydroxycholesterol, a potent ligand for the receptor EBI2/GPR183. This metabolite is particularly relevant to immune-cell migration and positioning. Another pathway involves sulfation by SULT2B1b, generating sulfated oxysterol species with distinct biological properties. Therefore, when interpreting the biology of 25-HC, it is often important to distinguish effects mediated by 25-HC itself from those mediated by its downstream metabolites (Nguyen et al., 2023).
3. Molecular Mechanisms of 25-Hydroxycholesterol
The biological influence of 25-HC cannot be reduced to a single receptor or pathway. Instead, it acts through multiple layers of regulation that include classical sterol-sensing systems, transcriptional control, membrane remodeling, lysosomal signaling, and metabolic reprogramming. This multi-level action helps explain why the same molecule can produce markedly different outcomes in distinct disease contexts.
3.1 Regulation of Cholesterol Homeostasis by 25-HC
One of the best-established actions of 25-HC is suppression of the SREBP pathway, especially SREBP-2, which governs cholesterol biosynthesis and uptake. 25-HC promotes retention of the SCAP-SREBP complex in the endoplasmic reticulum by favoring interaction with INSIG proteins, thereby preventing proteolytic activation of SREBP. As a result, transcription of genes involved in cholesterol synthesis and LDL uptake is reduced. This mechanism allows 25-HC to act as a feedback regulator that restrains intracellular cholesterol accumulation (Nguyen et al., 2024).
25-HC also intersects with the LXR pathway, although its role here is context-dependent and sometimes less dominant than that of other oxysterols. Through LXR-related signaling, 25-HC can promote expression of cholesterol transporters such as ABCA1, facilitating cholesterol efflux and contributing to reverse cholesterol transport. In inflamed tissues, this sterol-reprogramming activity is biologically important because cholesterol metabolism is intimately linked to innate immune signaling, membrane microdomain organization, and cytokine output (Nguyen et al., 2024).
3.2 Membrane Remodeling and Immunometabolic Signaling
25-HC also regulates cell function through non-canonical mechanisms that extend beyond transcriptional sterol control. Because it changes membrane accessibility of cholesterol and perturbs lipid raft organization, 25-HC can influence receptor trafficking, membrane fusion, endocytosis, and virus-cell entry. These physical effects on membranes help explain the broad antiviral activity described for many enveloped viruses, as well as some anti-inflammatory effects in selected cell systems (Zhang et al., 2023).
A major advance came from recent work showing that in immunosuppressive macrophages, 25-HC can accumulate in lysosomes and compete with cholesterol for binding to GPR155, thereby suppressing mTORC1 activity and activating AMPKα. This rewires macrophage metabolism and enhances STAT6-dependent immunosuppressive programming, including ARG1 expression. Mechanistically, this is important because it moves 25-HC biology beyond the classical SREBP/LXR paradigm and identifies it as a regulator of lysosomal nutrient sensing and macrophage cell fate (Xiao et al., 2024).
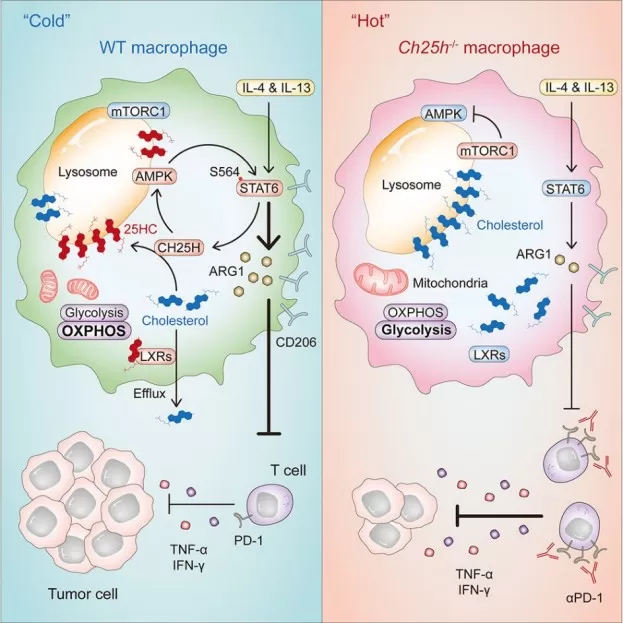
Image reproduced from Xiao et al., 2024, Immunity.
4. Dual Roles of 25-Hydroxycholesterol in Inflammation
Few aspects of 25-HC biology are as important, or as confusing, as its dual role in inflammation. In some settings it suppresses excessive inflammatory signaling and protects tissues from immune-mediated damage. In others it amplifies cytokine production, promotes inflammasome activation, enhances leukocyte recruitment, or supports pathogenic macrophage states. This apparent contradiction is not incidental. It reflects genuine context dependency shaped by cell type, dose, timing, disease stage, and metabolic background.
4.1 Anti-Inflammatory and Immunoregulatory Effects
A substantial body of work supports anti-inflammatory functions for 25-HC. By suppressing SREBP-driven lipid synthesis and altering intracellular cholesterol handling, 25-HC can limit activation of inflammatory programs that depend on sterol accumulation and membrane remodeling. Earlier studies established that CH25H-derived oxysterols can restrain NLRP3 inflammasome activity in some contexts and reduce IL-1β production, particularly when excessive cholesterol-driven inflammatory priming is involved. Recent reviews continue to recognize this protective side of the pathway, especially in settings where 25-HC serves as a homeostatic brake on lipid-driven innate activation (Nguyen et al., 2024; Carre et al., 2025).
25-HC can also dampen certain inflammatory signaling events by modifying membrane structure. In microglia, for example, 25-HC was shown to suppress IFN-γ-driven inflammatory responses by disrupting lipid raft formation and caveolin-dependent signaling compartments. Although this particular study predates 2022, it remains mechanistically relevant because it illustrates how 25-HC can act independently of nuclear receptors simply by reorganizing membrane sterol availability (Lee et al., 2022).
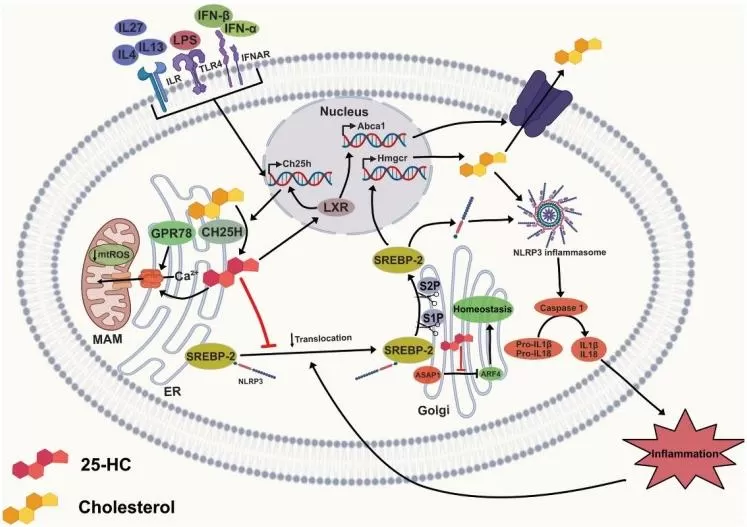
Image reproduced from Carre et al., 2025, Current Opinion in Endocrine and Metabolic Research, licensed under the Creative Commons Attribution 4.0 International License (CC BY 4.0).
4.2 Pro-Inflammatory and Tissue-Damaging Effects
At the same time, more recent work has made it clear that 25-HC can also act as a pro-inflammatory amplifier. The 2025 review by Carre and colleagues emphasizes that 25-HC and its downstream oxysterols are linked to mitochondrial dysfunction, endoplasmic reticulum stress, inflammasome signaling, cell death pathways, and neuroinflammation. In the central nervous system, this pro-inflammatory role is particularly well supported by new in vivo evidence (Carre et al., 2025).
In a 2024 open-access study, Romero and colleagues showed that peripheral LPS administration in mice induced brain CH25H expression and increased 25-HC levels. Higher 25-HC was associated with stronger cytokine production, microglial activation, and leukocyte infiltration, whereas genetic Ch25h deficiency attenuated these responses. The findings support a model in which 25-HC is not merely a bystander in neuroinflammation but an active driver of inflammatory amplification in the brain (Romero et al., 2024).
The same direction is seen in tauopathy. In a 2024 Journal of Experimental Medicine study, loss of Ch25h reduced age-dependent neurodegeneration and neuroinflammation in a mouse model expressing mutant human tau, with transcriptomic data showing broad suppression of pro-inflammatory microglial signaling. Together, these results argue that in chronic neurodegenerative settings, the Ch25h/25-HC axis may be pathogenic rather than protective (Toral-Rios et al., 2024).
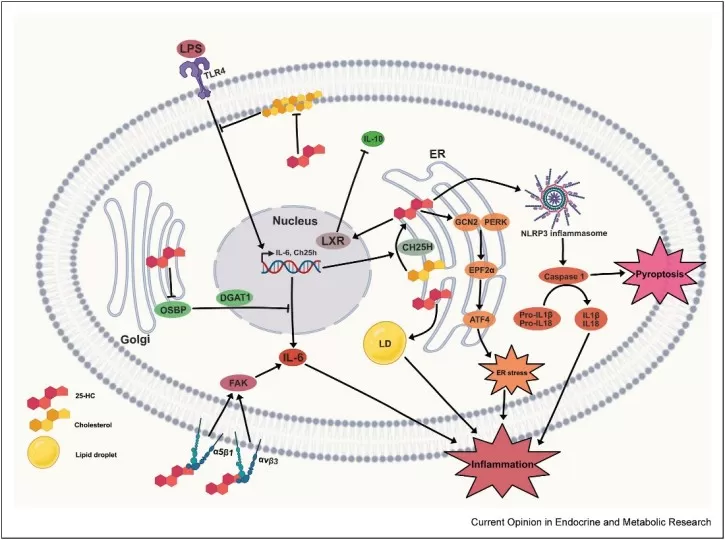
Image reproduced from Carre et al., 2025, Current Opinion in Endocrine and Metabolic Research, licensed under the Creative Commons Attribution 4.0 International License (CC BY 4.0).
5. Roles of 25-Hydroxycholesterol in Major Diseases
Because 25-HC regulates both lipid metabolism and immunity, its disease relevance extends well beyond one organ system. It appears in infectious disease, cardiovascular pathology, tumor immunology, neurodegeneration, and autoimmunity. What links these diverse conditions is not a single phenotype, but the ability of 25-HC to reshape local immune-metabolic states.
5.1 25-HC in Viral Infection and Antiviral Defense
25-HC has been widely studied as an antiviral lipid because CH25H is an interferon-stimulated gene and because 25-HC can alter membrane cholesterol accessibility required for viral fusion and entry. A 2023 Frontiers review summarized extensive evidence supporting antiviral activity against a range of enveloped viruses and highlighted mechanisms involving cholesterol redistribution, autophagy regulation, inflammatory signaling, and post-translational pathways. These findings help explain why the CH25H–25-HC axis is often presented as a component of intrinsic host defense (Zhang et al., 2023).
That said, antiviral activity does not automatically translate into therapeutic benefit in every infection model. In SARS-CoV-2-related settings, the literature suggests that the effect of 25-HC may be more nuanced than initially expected, with potential differences between early cell-based entry inhibition and later in vivo inflammatory injury. For translational work, this means 25-HC should be viewed less as a universally beneficial antiviral compound and more as a context-dependent lipid mediator whose net effect depends on timing, tissue compartment, and inflammatory burden (Nguyen et al., 2023; Zhang et al., 2023).
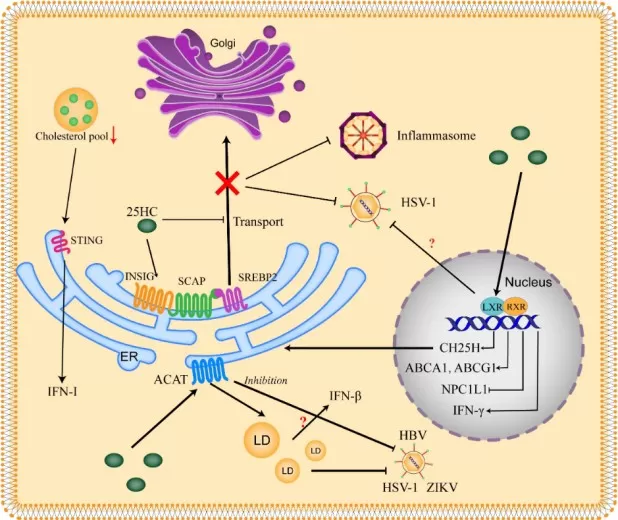
Image reproduced from Zhang et al., 2023, Frontiers in Immunology, licensed under the Creative Commons Attribution 4.0 International License (CC BY 4.0).
5.2 25-HC in Tumor Immunity and Cancer Progression
One of the most important recent advances in the field is the recognition of CH25H/25-HC as a regulator of tumor-associated macrophages (TAMs). In 2024, Xiao and colleagues showed that CH25H is induced in immunosuppressive macrophages by IL-4/IL-13 and tumor-associated signals. The resulting 25-HC accumulates in lysosomes, inhibits mTORC1 via GPR155 competition, activates AMPKα, and enhances STAT6-dependent immunosuppressive programming. Functionally, deleting CH25H in macrophages reduced immunosuppression, increased T-cell infiltration, and improved response to anti-PD-1 therapy, effectively helping convert "cold" tumors into more immune-responsive "hot" tumors (Xiao et al., 2024).
This work is especially important for drug discovery because it frames CH25H as an immunometabolic checkpoint, not just a metabolic enzyme. For oncology researchers, the implication is clear: 25-HC may contribute to tumor immune escape by stabilizing suppressive macrophage states. For translational platforms, it also raises the possibility that targeted oxysterol profiling could become relevant in tumor microenvironment studies and immunotherapy response assessment.
5.3 25-HC in Neuroinflammation and Neurodegenerative Disease
The CNS is another area where 25-HC is gaining importance. Recent work shows that CH25H is induced in inflammatory brain states and that loss of the pathway can reduce cytokine production, leukocyte infiltration, and neurodegenerative pathology in animal models. In tauopathy and LPS-induced neuroinflammation, the direction of evidence currently leans toward a pathogenic contribution of the Ch25h/25-HC axis, especially through microglial activation and pro-inflammatory signaling (Romero et al., 2024; Toral-Rios et al., 2024).
At the same time, downstream metabolism to 7α,25-dihydroxycholesterol connects 25-HC to EBI2/GPR183-dependent immune-cell positioning, which is highly relevant to neuroimmune trafficking and autoimmune disease. This means that part of the biological significance of 25-HC may come not only from its direct effects, but also from its ability to feed into oxysterol gradients that guide leukocyte behavior in inflamed tissues (Nguyen et al., 2024; Carre et al., 2025).
MetwareBio: Your Trusted Partner for 25-HC Analysis
As discussed above, 25-hydroxycholesterol (25-HC) is far more than a minor cholesterol oxidation product. It is a biologically active oxysterol with growing relevance in immunology, inflammation, viral infection, neurodegeneration, and cancer biology, largely because it links cholesterol metabolism to immune signaling and disease-associated lipid remodeling.
In research settings, accurate measurement of 25-HC is essential for clarifying pathway activity and biological function, yet this remains technically challenging. Since 25-HC is typically present at low abundance and can be generated artifactually during sample collection and preparation, reliable analysis depends on strict pre-analytical control, high analytical selectivity, and robust quantitative workflows. For this reason, LC–MS/MS has become the preferred strategy for 25-HC analysis, offering the sensitivity and specificity needed to distinguish low-level oxysterols from complex sterol backgrounds and structurally related metabolites.
To support this type of research, MetwareBio's Steroid Hormones Targeted Metabolomics Service provides a practical and pathway-oriented solution for absolute quantification of 40 steroid hormone-related compounds, covering key classes such as estrogens, androgens, progestogens, cholesterol, 25-hydroxycholesterol, and 7α,25-dihydroxycholesterol. Rather than measuring 25-HC in isolation, this panel enables researchers to profile it together with biologically connected sterol metabolites, improving mechanistic interpretation in studies of inflammation, endocrine regulation, metabolic disorders, and disease-associated signaling.
To ensure analytical reliability, our workflow is supported by 40 external standards and 23 internal standards, enabling robust quantitative performance across complex biological matrices. We use a high-performance LC–MS/MS platform with detection sensitivity at the ng/mL level, together with rigorous quality control procedures to support data accuracy and reproducibility.
If you are interested in steroid hormone profiling or 25-HC-related targeted analysis, please do not hesitate to contact us for more details.
Contact UsReferences
- Nguyen, C., Saint-Pol, J., Dib, S., Pot, C., & Gosselet, F. (2024). 25-Hydroxycholesterol in health and diseases. Journal of Lipid Research, 65(1), 100486. https://doi.org/10.1016/j.jlr.2023.100486
- Zhang, J., Zhu, Y., Wang, X., & Wang, J. (2023). 25-hydroxycholesterol: an integrator of antiviral ability and signaling. Frontiers in Immunology, 14, 1268104. https://doi.org/10.3389/fimmu.2023.1268104
- Romero, J., Toral-Rios, D., Yu, J., Paul, S. M., & Cashikar, A. G. (2024). 25-hydroxycholesterol promotes brain cytokine production and leukocyte infiltration in a mouse model of lipopolysaccharide-induced neuroinflammation. Journal of Neuroinflammation, 21(1), 251. https://doi.org/10.1186/s12974-024-03233-1
- Xiao, J., Wang, S., Chen, L., Ding, X., Dang, Y., Han, M., … & Wang, H. (2024). 25-Hydroxycholesterol regulates lysosome AMP kinase activation and metabolic reprogramming to educate immunosuppressive macrophages. Immunity, 57(5), 1087–1104.e7. https://doi.org/10.1016/j.immuni.2024.03.021
- Carre, R., Vigne, S. & Pot, C. (2025). 25-hydroxycholesterol in inflammation. Current Opinion in Endocrine and Metabolic Research, 40, 100582. https://doi.org/10.1016/j.coemr.2025.100582
- Toral-Rios, D., Long, J. M., Ulrich, J. D., Yu, J., Strickland, M. R., Han, X., … & Paul, S. M. (2024). Cholesterol 25-hydroxylase mediates neuroinflammation and neurodegeneration in a mouse model of tauopathy. The Journal of Experimental Medicine, 221(4), e20232000. https://doi.org/10.1084/jem.20232000
- Lee, J. H., Han, J. H., Woo, J. H., & Jou, I. (2022). 25-Hydroxycholesterol suppress IFN-γ-induced inflammation in microglia by disrupting lipid raft formation and caveolin-mediated signaling endosomes. Free Radical Biology & Medicine, 179, 252–265. https://doi.org/10.1016/j.freeradbiomed.2021.11.017


